Predictions & Data for this entry
| Model: abj | climate: Aw, Cfa | migrate: Mda, Ma | phylum: |
| COMPLETE = 2.9 | ecozone: TPi, THp | food: biCi | class: |
| MRE = 0.089 | habitat: 0jFm, jiMm | gender: D | order: |
| SMSE = 0.018 | embryo: Fs, Fh | reprod: O | family: |
Zero-variate data
| Data | Observed | Predicted | (RE) | Unit | Description | Reference |
|---|---|---|---|---|---|---|
| ab | 11 | 11.78 | (0.07059) | d | age at birth | Fabe2013 |
| tj | 28 | 25.25 | (0.0981) | d | time since hatch at metamorphosis | Fabe2013 |
| ap | 60 | 58.9 | (0.01833) | d | time since hatch at puberty | Fabe2013 |
| am | 2008 | 2008 | (0.000123) | d | life span | EgamEtoh1969 |
| Lb | 0.45 | 0.3961 | (0.1197) | cm | total length at birth | Iwam2004 |
| Lj | 1.3 | 1.212 | (0.06774) | cm | total length at metamorphosis | Iwam2004 |
| Lp | 2.53 | 2.568 | (0.01522) | cm | total length at puberty | KangYoko2002 |
| Li | 4 | 4.211 | (0.0527) | cm | ultimate total length | Fabe2013 |
| Wd0 | 0.00024 | 0.0002059 | (0.1421) | g | egg dry weight | Stan1972 |
| Wwb | 0.00065 | 0.0006432 | (0.01047) | g | wet weight at birth | MartHint1995 |
| Wdb | 0.00013 | 0.0001286 | (0.01047) | g | dry weight at birth | MartHint1995 |
| Wwp | 0.178 | 0.1753 | (0.01495) | g | wet weight at puberty | KangYoko2002 |
| Wwi | 0.8 | 0.7726 | (0.03429) | g | ultimate wet weight | AnklJohn2004, GhonEgam1982 |
| Ri | 30 | 32.1 | (0.07007) | #/d | maximum reprod rate | AnklJohn2004 |
Uni- and bivariate data
| Data | Figure | Independent variable | Dependent variable | (RE) | Reference |
|---|---|---|---|---|---|
| tW_f | 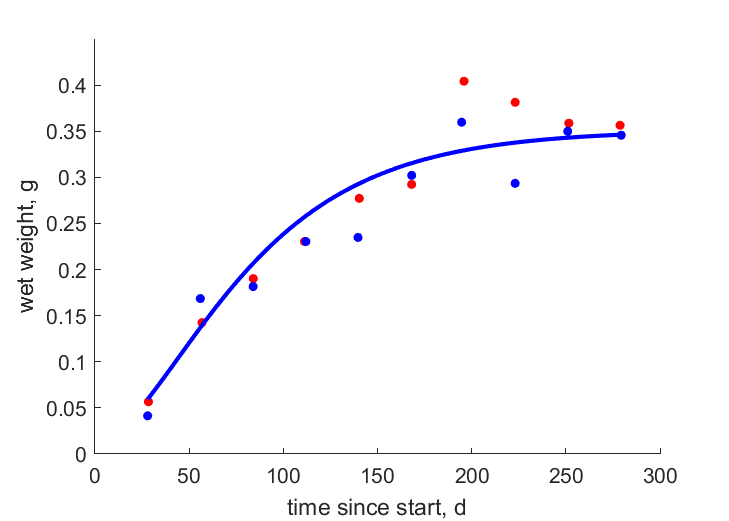  | time since start | wet weight | (0.0869) | TehHint1998 |
| tW_m |   | time since start | wet weight | (0.1021) | TehHint1998 |
| tN1 | 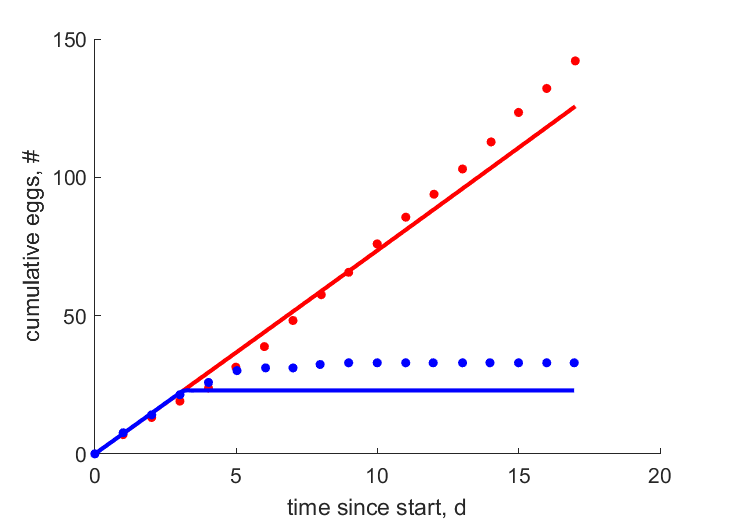  | time since start | cumulative eggs | (0.08362) | HaseKand2016 |
| tN0 |   | time since start | cumulative eggs | (0.2467) | HaseKand2016 |
| tW1 | 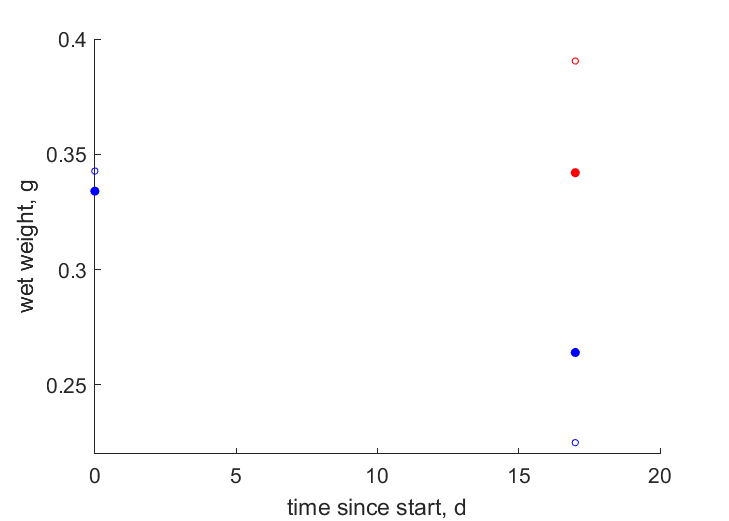  | time since start | wet weight | (0.08459) | HaseKand2016 |
| tW0 |   | time since start | wet weight | (0.08069) | HaseKand2016 |
| tL | 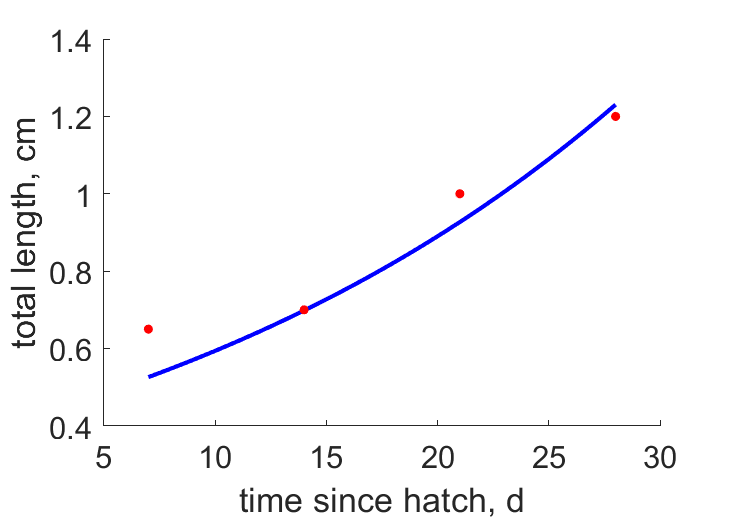 | time since hatch | total length | (0.06461) | SpenHuss2006 |
| tL2 | 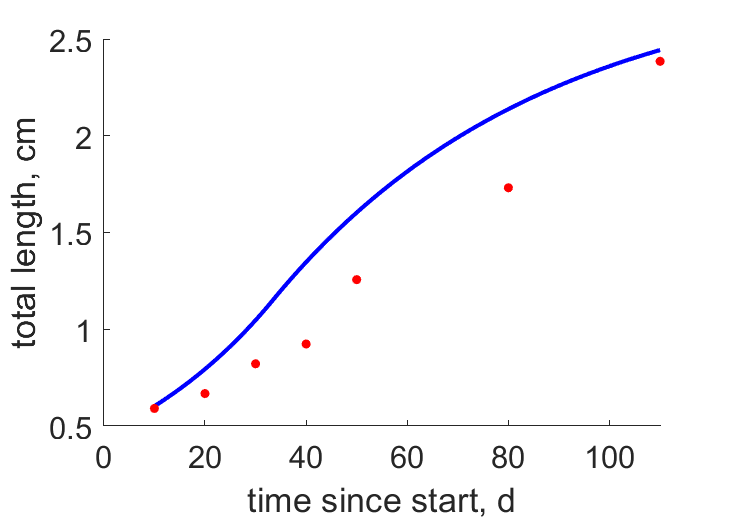 | time since start | total length | (0.1909) | GonzCarb2016 |
| tS | 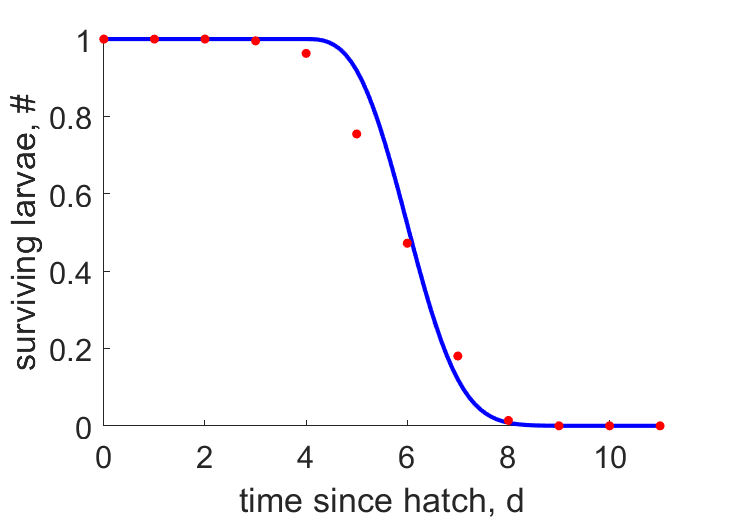 | time since hatch | surviving larvae | (0.0502) | Stan1972 |
| tW | 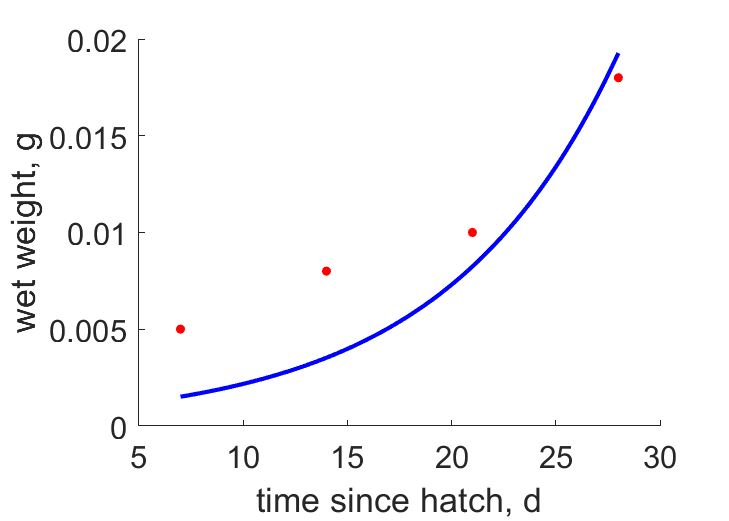 | time since hatch | wet weight | (0.2684) | SpenHuss2006 |
| tW2 | 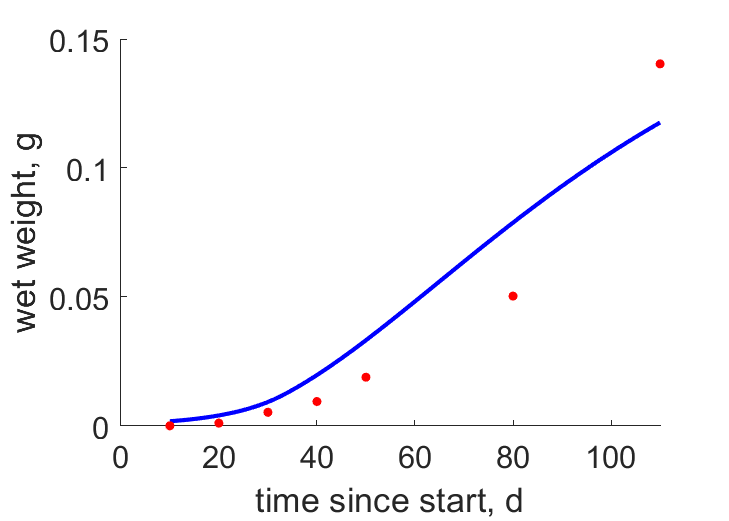 | time since start | wet weight | (0.3755) | GonzCarb2016 |
Pseudo-data at Tref = 20°C
| Data | Generalised animal | Oryzias latipes | Unit | Description |
|---|---|---|---|---|
| v | 0.02 | 0.01056 | cm/d | energy conductance |
| kap | 0.8 | 0.4705 | - | allocation fraction to soma |
| kap_R | 0.95 | 0.95 | - | reproduction efficiency |
| p_M | 18 | 364 | J/d.cm^3 | vol-spec som maint |
| k_J | 0.002 | 0.002 | 1/d | maturity maint rate coefficient |
| kap_G | 0.8 | 0.8033 | - | growth efficiency |
Discussion
- Contrary to the version 2017/09/22: Males and females have same parameters
- Starvation data is included in early lifes stages: modelled using eqn in AuguLitv2011.The difference is that, once a shrinking threshold del_X is surpassed,the hazard from shrinking is taken proportional to the difference between the amount of structure left and the amount of structure before shrinking started.
- Starvation data for adults is included: spawning is continuous.Buffer is emptied at each spawning event.Spawning ceases when energy mobilised from reserve no longer covers somatic maintenance andEnergy from repro buffer is first used to cover som main. (AuguGagn2012).If no energy is left in the repro buffer then energy from structure.The initial struture and initial amount in the repro buffer are free parameters.
- The interpretation of some data was revised with respect to version 2017/09/22.TehHint1998: age of fish at start of experiment (time 0) is 25 days post hatch. This information is now implemented in the predict file. GoncCarb2016: average age of fish at start of experiment (time 0) is 5.5 days post hatch. This information is now implemented in the predict file. Wwi: there was an error in 2017/09/22. the male and female Wwi were inversed. In this version we used the Wwi for females from a more recent study and no longer include different parameters for males as there is not enough data.Shape coefficients: contrary to 2017/09/22 there is only one shape coefficient which applies for total length as all length data are total length.There are no longer different shape coefficients for different life-stages.The egg dry weight is included.
Bibliography