

| email: | gineke@bio.vu.nl |
| room: | B402 |
| phone: | +31-20-44.47135 |
| Project: | Physiologically based toxicokinetics |
One of the tasks of The National Institute of Public Health and Environmental Protection (RIVM) in Bilthoven is risk assessment.
In risk assessment there is no international agreement about norms and measures. For example for dioxin the acceptable daily intake (ADI) (for humans) proposed by the World Health Organization (WHO) differs by a factor 103 from the ADI proposed in the United States.
These dissimilarities are mainly caused by differences in extrapolation from dose-effect relations for test-animals to ADI values for humans. Usually the highest (daily) dose for which no effect on the (most sensitive) animal species is observed (No Observed Effect Level, NOEL) is divided by a safety factor for obtaining the ADI value. The safety factor usually consists of a factor to account for intraspecies variation since humans might be more sensitive to the toxicant and a factor to account for variation between individuals, because the human population contains more variation than the group of tested animals. Other ways of intraspecific scaling are also being used. For genotoxicological carcinogens (toxicants which cause DNA damage) the NOEL is assumed to be zero. For these compounds the ADI is set using the probability of harmful effects.
To dispose of the arbitrariness in the extrapolation ways of estimating risk which are based on mechanisms are needed. A model which links intake with internal concentrations will contribute to this objective. The aim of this project is the development of such a model for lipophilic compounds, and the estimation of the parameter values to be used in this model.
We have developed a physiologically based pharmacokinetic model to describe the uptake, distribution and elimination of toxicant in humans during a life-time. The model contains age-dependent physiological parameters that can account for interspecies variation, but do not depend on the toxicant. This model is linked to a model that describes the age dependent intake rates of dioxins and furans for the entire (Dutch) population. Elimination rate depends on the congener.
Model simulations have shown that historical changes in intake, due to changes in environmental concentrations and in dietary habits, greatly influence 2,3,7,8-TCDD concentrations in persons with background intake (Fig 1).
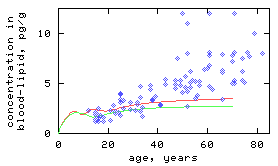
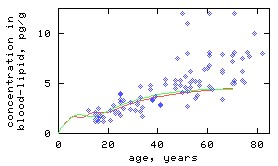
Figure 1
Red curve = male; green curve = female; blue dots = cross-section through the German population.
- left Model simulation of a single person with an elimination rate constant of 15.6 (per year).
- right Simulated cross-section of the population, with an elimination rate constant of 15.6 (per year) in males, and 12.0 (per year) in females and a correction function for time dependent changes in the intake.
At the moment we are collaborating with Dr. J.E. Michalek from the Air Force Health Study in the United States. We estimated the elimination rate of 2,3,7,8-TCDD from his concentration measurements in Vietnam Veterans who were previously exposed to high concentrations.
We are also collaborating with the group of Prof. Selenka at the Ruhr University Bochum, who measured concentrations of several dioxin and furan congeners in a cross section of the German population. We are currently estimating the elimination rates of all measured congeners from these data. It appears that the elimination rate estimates from these data are very sensitive to the intake rates.
In certain test-animals high doses of toxicant not only affect health directly, but also affect growth (or bodyweight). This might be caused by the fact that elimination of the toxicant costs a considerable amount of energy. This aspect will be looked into with use of the Dynamic Energy Budget model (DEB) that has been developed by Kooijman (in this departement).